Draw The Electron Configuration For A Neutral Atom Of Nickel
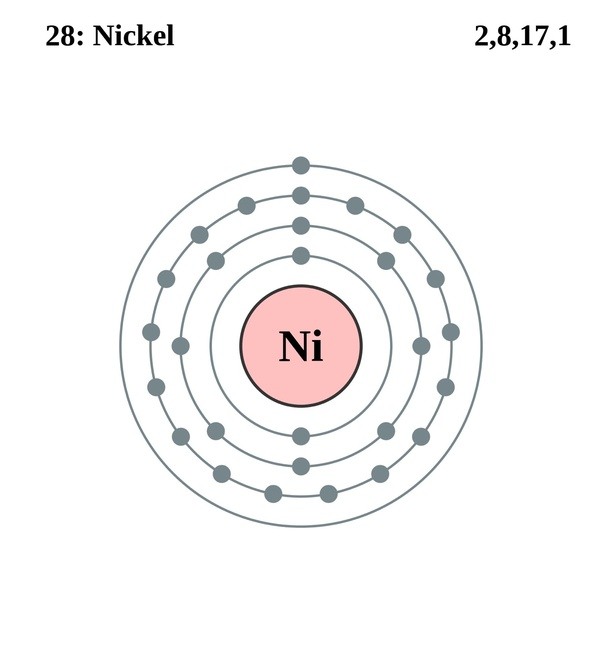
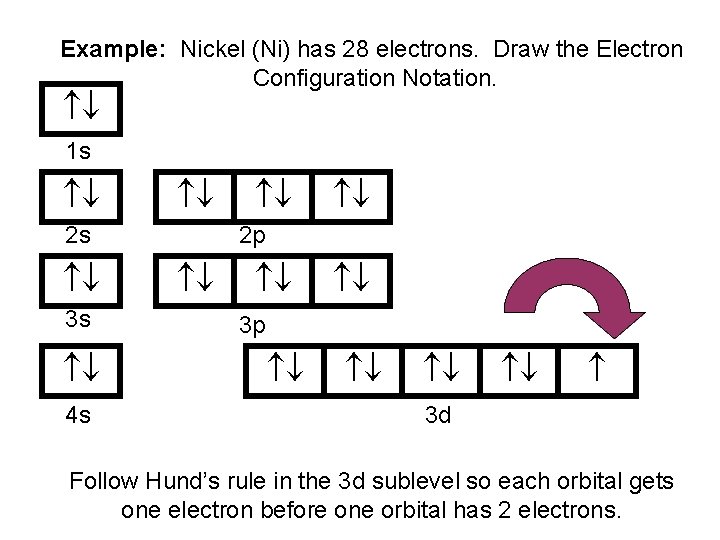
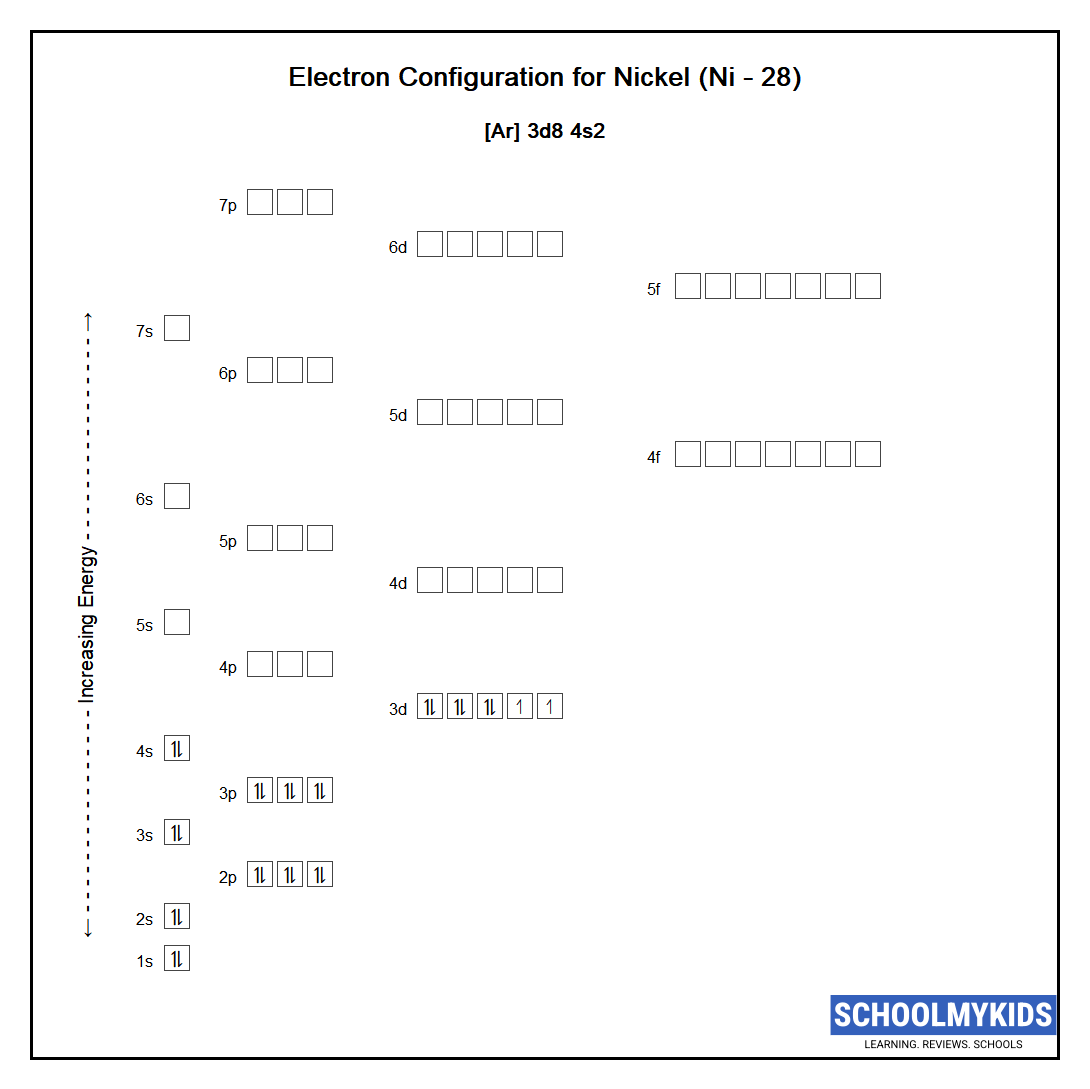
Draw The Electron Configuration For A Neutral Atom Of Nickel - The first three (n, l, and m l) may be the same,. The pauli exclusion principle states that no two electrons can have the same four quantum numbers. We first need to find the number of. We write electronic configurations by following the aufbau principle (from german, meaning “building up”). Web the ground state electron configuration of a neutral nickel atom is #[ar]3d^84s^2#.this data comes from the nist atomic spectra database. The neutral nickel atom therefore must have 2 8 electrons to accommodate according to the usual. Web a nickel nucleus has 28 positively charged nuclear particles, 28 protons. Web the ground state electron configuration of nickel is [ar].3d8.4s2. Following the electron configuration rules, such. Web assigning electron configuration. Mastering the electron configuration of nickel ([ar] 3d^8 4s^2) reveals vital insights into its atomic structure. Web ni electronic configuration: Web the ground state electron configuration of nickel is [ar].3d8.4s2. We first need to find the number of. 1s 2 2s 2 2p 6 3 s 2 3p 6 4s 2 3d 3; Web nickel is in the #4th# energy level, #d# block, #7th# column, this means that the electron configuration will end #3d^8# with the #d# orbital being one level lower. 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d⁸ so, the electron configuration for a neutral atom of nickel is $\boxed{1s^2 \ 2s^2 \ 2p^6 \. Determine how many electrons were lost. The presence of 10 valence electrons in nickel contributes to its unique chemical properties. Web the arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We write electronic configurations by following the aufbau principle (from german, meaning “building up”). Mastering the electron configuration of nickel ([ar] 3d^8 4s^2) reveals vital insights into its atomic structure. We describe an electron configuration with a symbol that. Following the electron configuration rules, such. Web write the configuration of the neutral atom. Web the same rule will apply to transition metals when forming ions. We first need to find the number of. To find the electron configuration. The neutral nickel atom therefore must have 2 8 electrons to accommodate according to the usual. 1s 2 2s 2 2p 6 3 s 2 3p 6 4s 2 3d 3; Web to write an electron configuration for a cation, start by writing the electron configuration for the neutral atom. Web electron configurations describe where electrons are located around the nucleus of an atom. Web write the configuration of the neutral atom. Web a nickel nucleus has 2 8 positively charged nuclear particles, 2 8 protons. Mastering the electron configuration of. For example, the electron configuration of lithium, 1s²2s¹,. Web to write the configuration for the nickel ions, first we need to write the electron configuration for just nickel (ni). Web a nickel nucleus has 28 positively charged nuclear particles, 28 protons. The presence of 10 valence electrons in nickel contributes to its unique chemical properties. The neutral nickel atom therefore. Web a nickel nucleus has 2 8 positively charged nuclear particles, 2 8 protons. Web the ground state electron configuration of nickel is [ar].3d8.4s2. The neutral nickel atom therefore must have 28 electrons to accommodate according. Web to write the configuration for the nickel ions, first we need to write the electron configuration for just nickel (ni). The presence of. Web write the configuration of the neutral atom. Following the electron configuration rules, such. Web electron configurations describe where electrons are located around the nucleus of an atom. The first three (n, l, and m l) may be the same,. Web the ground state electron configuration of a neutral nickel atom is #[ar]3d^84s^2#.this data comes from the nist atomic spectra. The presence of 10 valence electrons in nickel contributes to its unique chemical properties. Web the ground state electron configuration of nickel is [ar].3d8.4s2. Mastering the electron configuration of nickel ([ar] 3d^8 4s^2) reveals vital insights into its atomic structure. Determine how many electrons were lost. Web the same rule will apply to transition metals when forming ions. Web click here:point_up_2:to get an answer to your question :writing_hand:how do you write the electron configuration for nickel. Web the ground state electron configuration of nickel is [ar].3d8.4s2. The pauli exclusion principle states that no two electrons can have the same four quantum numbers. The first three (n, l, and m l) may be the same,. Web assigning electron configuration. Web to write the configuration for the nickel ions, first we need to write the electron configuration for just nickel (ni). The presence of 10 valence electrons in nickel contributes to its unique chemical properties. Web a nickel nucleus has 2 8 positively charged nuclear particles, 2 8 protons. [ar], 3d8, 4s2 atomic number of nickel (ni) = 28 electronic. Web the ground state electron configuration of nickel is [ar].3d8.4s2. Mastering the electron configuration of nickel ([ar] 3d^8 4s^2) reveals vital insights into its atomic structure. Web to write the configuration for the nickel ions, first we need to write the electron configuration for just nickel (ni). The first three (n, l, and m l) may be the same,. Web. Web to write an electron configuration for a cation, start by writing the electron configuration for the neutral atom. Determine how many electrons were lost. The first three (n, l, and m l) may be the same,. Web the electron configuration for nickel is: Web click here:point_up_2:to get an answer to your question :writing_hand:how do you write the electron configuration for nickel. We write electronic configurations by following the aufbau principle (from german, meaning “building up”). Then determine the number of electrons. Web a nickel nucleus has 2 8 positively charged nuclear particles, 2 8 protons. To find the electron configuration. Mastering the electron configuration of nickel ([ar] 3d^8 4s^2) reveals vital insights into its atomic structure. Web the arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. The neutral nickel atom therefore must have 2 8 electrons to accommodate according to the usual. Web assigning electron configuration. [ar], 3d8, 4s2 atomic number of nickel (ni) = 28 electronic configuration is 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d8 or [ar], 3d8, 4s2 where. For example, the electron configuration of lithium, 1s²2s¹,. The neutral nickel atom therefore must have 28 electrons to accommodate according.Nickel atomic structure has atomic number, atomic mass, electron
Nickel Electron Configuration (Ni) with Orbital Diagram
Nickel Electron Configuration(Explained for Beginners)
Symbol and electron diagram for nickel Royalty Free Vector
Nickel(Ni) electron configuration and orbital diagram
Electron Configuration For Nickel cloudshareinfo
Nickel Atom Science Notes and Projects
Electron Configuration For Nickel cloudshareinfo
Electron Configuration Chapter 5 Electrons have 3 levels
Nickel (Ni) Element Information, Facts, Properties, Uses Periodic
Web The Ground State Electron Configuration Of A Neutral Nickel Atom Is #[Ar]3D^84S^2#.This Data Comes From The Nist Atomic Spectra Database.
1S² 2S² 2P⁶ 3S² 3P⁶ 4S² 3D⁸ So, The Electron Configuration For A Neutral Atom Of Nickel Is $\Boxed{1S^2 \ 2S^2 \ 2P^6 \.
We Describe An Electron Configuration With A Symbol That.
Web The Ground State Electron Configuration Of Nickel Is [Ar].3D8.4S2.
Related Post: